|
7/23/2023 0 Comments Water emulsion meaning
In W/O emulsions, simple steric effect is the key role for stabilizing the system, owing to the low electrical conductivity of the water (continuous phase) and their products can be in the solid or semi-solid and liquid forms. The single emulsion is a colloidal dispersion of two immiscible liquids (water and oil) and is simply divided into two systems as oil-in-water (O/W) or water-in-oil systems (W/O) depending on the dispersed phase (oil) or continuous phase (water). Įmulsions can be classified according to the structure of the phases as single, multiple emulsions (a size range 0.1–5 μm), micellar emulsions, or micro-emulsions (a size range of 5–50 nm) and nano-emulsions (a size range of 10–500 nm). Synthetic surfactants and biosurfactants are two basic groups. Stabilizers decrease interfacial tension and conversely increase the surface area of immiscible phases. 
Stabilizers are largely biopolymers (proteins, polysaccharides, phospholipids, plant-based emulsifiers) and are amphiphilic molecules which contain hydrophobic and hydrophilic parts. Emulsifiers create special area for preventing them from aggregation and having low HLB value. They enhance its stability by reducing the interfacial tensions of dispersed phase-continuous phase (oil–water), and the van der Waals’ steric and electrostatic repulsion have significant roles in stabilization. Emulsifiers are small surface-active molecules and contain hydrophilic and hydrophobic areas, so they change the structure of the interface. The stability of the system is related to the empirical hydrophile–lipophile balance (HLB) number of the emulsion. The stability of emulsion depends on the steric hindrance and electrostatic interactions and the viscosity of the continuous phase (gelation). 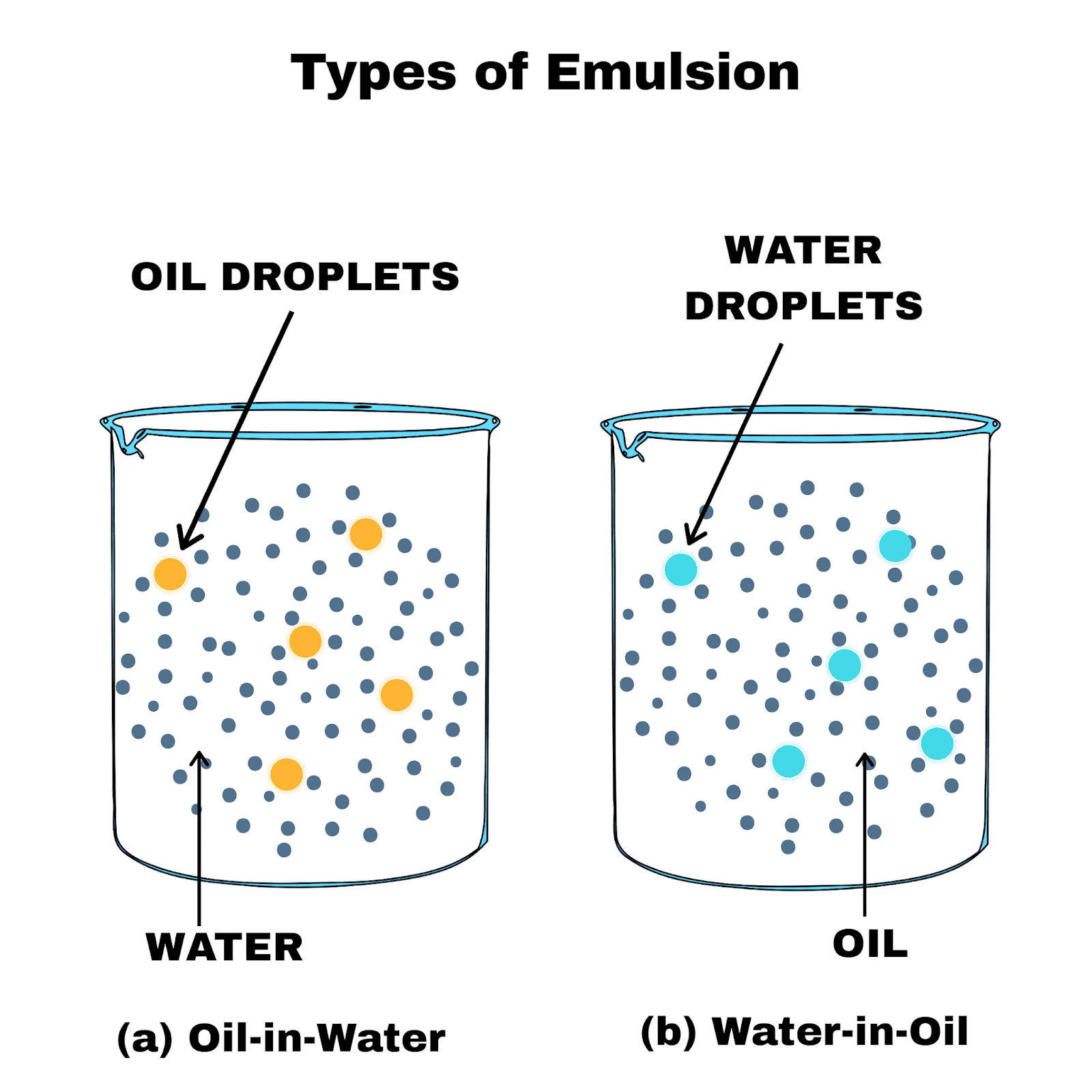
The average particle size and its distribution are crucial factors for stability of emulsion which depends on the rate of coalescence and also other important factors are the aggregation of the droplets, ionic strength, concentration, temperature, pH, energy, osmotic pressure, viscosity, interfacial tension and dynamically the addition of emulsifying agent (emulsifier), and stabilizer. Emulsions are thermodynamically unstable (coalescence, sedimentation/creaming, flocculation, Ostwald ripening, and phase inversion) and are provided with kinetic stability by surfactant molecules for weeks, months, or years.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
